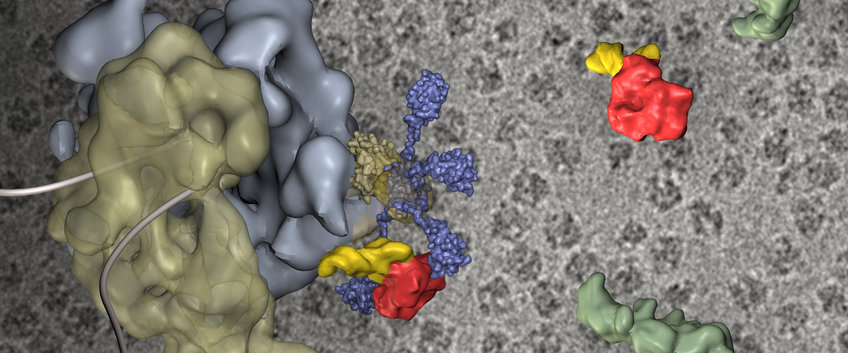
Structural Dynamics
The aim of our group is to determine three-dimensional structures of macromolecules with the single particle electron cryomicroscopy technique. Our work is mainly focused on spliceosomal components called snRNPs to obtain structural information about the eucaryotic pre-mRNA splicing. Apart from that we are working on ribosomal, viral and oxygen carrier projects. More detailed information is provided on our group website.
Press Releases & Research News
Researchers led by Ashwin Chari and Holger Stark have visualised the structure of this fatty acid factory at an unprecedented resolution of 1.9 Å. At this level of detail, they were able to directly observe enzyme reactions and track a complete fatty acid production cycle. The findings provide new approaches for combating pathogens and producing fatty acids sustainably.
more
Holger Stark and his team have broken a crucial resolution barrier in cryo-electron microscopy. The scientists succeeded in observing single atoms in a protein structure and taking the sharpest images ever with this method. Such unprecedented details are essential to understand how proteins perform their work or cause diseases in the living cell. The technique can in future also be used to develop active compounds for new drugs.
more
Show more
Macromolecular machines in 3D: The complex world of complexes Tiny nano machines called macromolecular complexes participate in the most fundamental biological processes. The high-resolution three-dimensional (3D) structure of these complexes and their dynamic behavior can be studied by cryo electron microscopy. The molecular movies that can be obtained for these nano machines contribute tremendously to our understanding of molecular processes at a structural level.
(in German)
more