Optical Nanoscopy in Neuroscience
Optical Nanoscopy
Optical light microscopy is widely used to visualize tiny structures. It has great potential especially for use in the life sciences, because of the possibility to image living cells or even small organisms. Cellular structures can be marked very specifically with a fluorescent dye and imaged with high contrast. With this technique any part of the cell such as the cytoskeleton, the nucleus or functional units such as receptors can be visualized. The spatial resolution of conventional far-field (fluorescence) light microscopy, however, is limited meaning that small details cannot be resolved. For example, the nuclear pore, a protein complex which allows the transport of molecules through the nuclear envelope, cannot be resolved with light microscopy because of its small size of about 120 nm.
This all changed two decades ago, as a novel light microscopy technique came of age, which revolutionized fluorescence microscopy by utterly breaking the diffraction barrier. In the nineties Prof. Dr. Stefan W. Hell (Max Planck Institute for Biophysical Chemistry, Göttingen) discovered and then pioneered STED microscopy, which was the first to resolve tiny structures with a far-field light microscope below the diffraction barrier. In brief, STED microscopy utilizes not only an excitation light beam, but also a second, tailored light beam, which is capable to switch off part of the fluorescence by STimulated Emission Depletion. Since the conception of STED microscopy a whole family of novel light microscopy methodologies have been developed, which are generalized under the general terms “nanoscopy” or “superresolution”.
Neuroscience, a fertile and challenging ground for STED microscopy
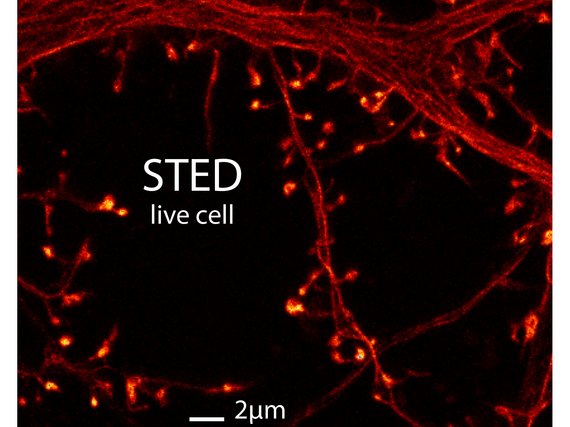
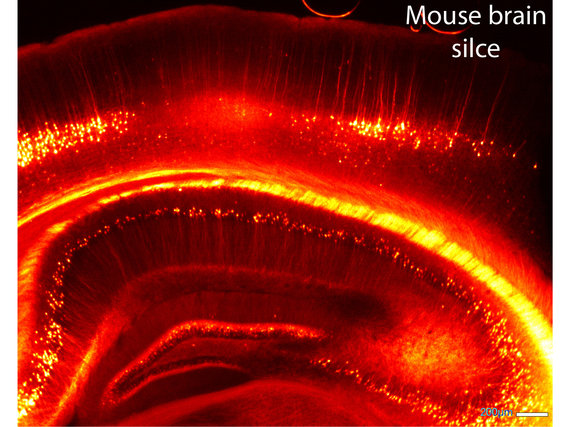
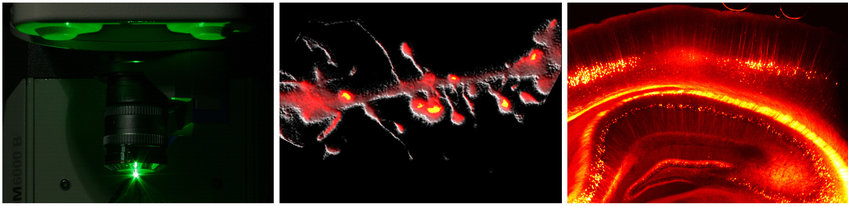
The contact sites between two neurons, called ‘synapses’, are the most fundamental information processing units in the brain. Excitatory synapses are frequently formed by dendritic spines, which are small protrusions of the neuronal dendritic arborizations and which are purported to be the smallest functional processing units in the central nervous system. The minute size of synapses creates an inherent difficulty for conventional imaging but makes them an ideal target for STED microscopy. Because even a single neuron establishes a huge amount of synapses, they are best observed in undamaged, intact tissue, such as in brain slices or even better in living animals. STED microscopy is ideally suited to study synaptic processes in living systems, because of several useful properties: of all the nanoscopy techniques, STED microscopy stands out for its fast recording speed, its inherent 3D sectioning capability, the potential to image deep within tissue, and the possibility of exploiting a vast array of commercially available fluorescent dyes.
In recent years we have used STED microscopy to image dendritic spines in living tissue. More specifically we have been examining the structural dynamics of dendritic spines, which are thought to form the basis of memory in the brain. We will continue to study structural changes and morphological details using nanoscopy, thereby helping to decipher how exactly our brains function.
Media (in German)
Deutschlandfunk: Dem Gehirn beim Lernen zusehen
Life Science Standort Niedersachsen, Video mit Blick in unser Labor.