Enhancing carbon-13 NMR signals in liquids
Limitations in sensitivity often complicate the analysis of complex (bio)molecules by nuclear magnetic resonance (NMR) spectroscopy. Now, a research team headed by Marina Bennati at the Max Planck Institute for Biophysical Chemistry, together with colleagues at the University of Florence (Italy), has shown that carbon-13 (13C) NMR signals can be strongly enhanced in solution by resonant microwave irradiation of a nitroxide organic radical used as polarizer for 13C nuclei. The new method shows up to 1000-fold improvements in sensitivity and promises to study small molecules and metabolites in much greater detail.
NMR is a fundamental spectroscopic technique for studies of biological systems and materials, molecular imaging as well as analytics of small molecules. Since it detects interactions at a very low energy scale, it is non-invasive and applications include animals and humans. Despite of its achievements, one of its most severe limitations is the low sensitivity, which results from the small interaction energies involved. Accordingly, this issue has been a long-standing goal in the field, leading the community to a continuous search for new excitation and detection technologies.
One way to boost NMR signals is through dynamic nuclear polarization (DNP), in which microwave irradiation transfers spin polarization from electrons of a stable organic radical to nuclear spins of interest. DNP has historically worked well in solid-state NMR experiments, but struggled in liquids. Marina Bennati and her team, in collaboration with Italian colleagues, have now demonstrated, for the first time, that signal enhancement of 2 to 3 orders of magnitude for room-temperature 13C NMR experiments can be achieved by DNP at magnetic fields around 3 Tesla and experimental conditions relevant for liquid NMR as well as magnetic resonance imaging (MRI).
Polarization transfer in liquids is driven by electron-nuclear cross relaxation resulting from molecular motion. It was thought that translational and rotational diffusion of molecules provide the main mechanism for cross relaxation, which becomes intrinsically ineffective at increasing magnetic fields (that is, at Larmor resonance frequencies of electron spins around 100 GHz). One work-around proposed in the past has been to polarize the nuclei at very low temperatures in the solid state and then rapidly warm up the sample into the liquid state. But that approach limits the signal acquisition to a single scan and poses the sample under an enormous temperature-stress. After optimizing several parameters in the DNP-NMR protocol, the scientists observed that fast molecular collisions in the pico- to sub-picosecond time scale can drive the polarization transfer at high magnetic fields by modulating scalar hyperfine interactions between electron and 13C nuclear spins.
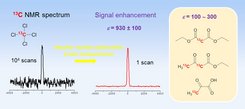
The figure illustrates the 13C NMR spectrum of 13CCl4 at 3.4 Tesla without (left) and with DNP (center). The spectrum without DNP required about 2 days of signal averaging, whereas the one with DNP takes only 15 seconds. The inset (right) shows representative organic compounds, for which considerable signal enhancements were achieved.
The experiment can be repeated within seconds for signal averaging without interfering with the sample magnetic homogeneity. Thereby, the method is compatible with conditions required in high-resolution NMR. Enhancement of 13C signals on various organic compounds, particularly on metabolites used for cancer imaging, opens up new perspectives for DNP as a general tool to increase sensitivity in solution NMR for metabolomics, structural and dynamic characterization of small biomolecules as well as a suitable tool in MRI.
The results now raise the need of much broader investigations such as screening of signal enhancements in several compounds as well as thinking about suited devices for larger sample volumes to avoid microwave absorption and heating in aqueous solutions. The present detection of a very efficient mechanism for NMR signal enhancements in liquids opens up new perspectives for applications of liquid NMR spectroscopy, which would benefit greatly from higher sensitivity. (MB)





